  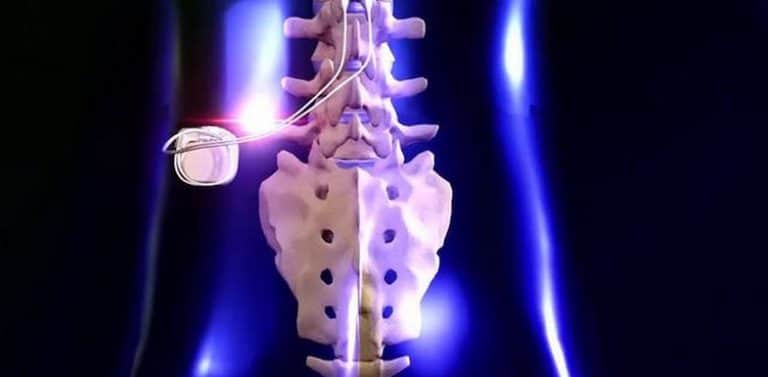
Labeling Change - Indications/instructions/shelf life/tradenameĪpproval for expanding the indications to include non-surgical back pain (NSBP) for the tonic and BurstDR stimulation modes, and diabetic peripheral neuropathy (DPN) of the lower extremities for the tonic stimulation mode, for the Prodigy, Proclaim XR, Proclaim Plus, and Externa Spinal Cord Stimulation (SCS) Systems. 24, 2022 /PRNewswire/ - Medtronic plc (NYSE:MDT), a global leader in healthcare technology, today announced it has received U.S. The device offers lower-dose and recharge-free neurostimulation for patients with chronic pain with a battery life for up to 10 years. Stimulator, spinal-cord, totally implanted for pain relief Abbott won US FDA approval for its Proclaim XR spinal cord stimulator. Prodigy, Proclaim XR, Proclaim Plus, and Externa Spinal Cord Stimulation (SCS) Systems Be sure to look at the original PMA record for more information. A supplement may have changed the device description/function or indication from that approved in the original PMA. At Abbott, we understand the heavy burden of chronic pain and how it can take over your life. 
The Infinity Deep Brain Stimulation system delivers mild pulses of electricity to the precise areas within the brain. 1 Each year about 50,000 people receive SCS therapy, a type of neurostimulation therapy. The Proclaim DRG Neurostimulation System is the first and only neurostimulation device approved only for complex regional pain syndrome (CRPS) and those seeking relief from causalgia -nerve pain following surgery or injury. Note: This medical device record is a PMA supplement. Spinal cord stimulation (SCS) therapy has been used for more than 55 years to manage chronic pain and improve quality of life. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
